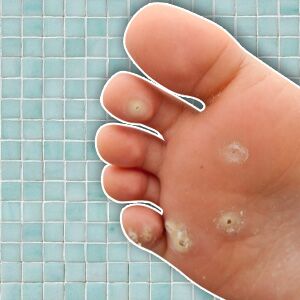
The Human Papillomavirus (HPV) causes warts. This is the most common type of viral infection on the skin. Plantar warts tend to grow where there is pressure and friction. So, the heels and soles of the feet make great growing grounds. A lot of these warts are very resistant to treatment; therefore, a medical consultation is necessary. Read more on a new unique and promising treatment for resistant plantar warts.
For resistant plantar warts, your doctor may try laser therapy, cryotherapy, cantharidin liquid, or other techniques. If these therapies fail, then another treatment is intralesional injections of Candida Albicans Antigen.
The doctor will give you a prescription for Non-active Candida Albicans Antigen to take to a pharmacist. A small glass vial is dispensed. Also, a numbing agent is added at our office for your comfort. You may want to take Tylenol or Ibuprofen before a visit to help with the discomfort.
During your doctor’s visit, the doctor injects a small amount of the dead yeast into as many warts as possible. It is not necessary to inject every wart; however, the doctor injects the largest first. As many as possible receive the injection limiting the total amount used to 1 cc.
The advantages are that it is quick, and there are no scars from the injections or open sores to deal with. So, you can immediately return to full activity, including swimming, sports, jazzercise, etc…. Also, no special care is needed. With injection therapy, the body’s immune system learns that wart tissue is abnormal starting an immunostimulatory effect. Your own immune cells break down the Wart Virus. Unlike other treatments, if the warts go away with injection therapy, they rarely return.

The side effects of injection therapy with Candida have been rare. Rarely someone will develop a rash, hives, itching over the whole body, or swelling. This indicates an allergy and will mean that the patient can no longer receive any further injections. Itching and redness in the area of injection are to be expected, however. Rarely, there will be some mild blistering. Often warts will turn somewhat black and the crust will fall off. About one-third of the time, a second injection is given one month later. Half of remaining warts will respond to the second injection.
If your warts show no response after the first two injections; you can receive a third injection one month later. (We do not normally give more than three monthly injections, but in some instances, we have given them up to five times.) 85% of patients will have cleared their warts after the third injection. With no results after the third injection, you should try another method.
If you notice a rash after treatment, please call our office as soon as possible. If you develop hives take Benadryl immediately and call our office. (50 mg for older children/100 mg for adults) For children less than 5 years old, check with us about the dosage.
Generally, a follow-up visit is scheduled for 3 weeks to one month after the first injection. Also, you can help by eating a diet rich in fruits and vegetables and taking a multivitamin. We’ll be glad to answer any questions you might have.
Of 11 patients who completed one study, 9 had complete resolution of their treated warts (82%), 1 had partial resolution(9%), and 1 had no response (9%). Complete resolution of the first distant untreated warts was observed in 6 of 8 patients(75%), while that of the second distant warts was observed in 6 of 6 patients (100%), during a phase 1 clinical trial.
According to the American Family Physician articles, Candidal Injections are a promising treatment for plantar warts.
Mulhem, E, Pinelis S. Treatment of Nongenital Cutaneous Warts. Am Fam Physican. 2011; 1;84 (3) 288-93
Kevin H. Kim, BS; Thomas D. Horn, MD; Joni Pharis, BBA; Jay Kincannon, MD; Robert Jones, MD; Kevin O’Bryan, MD; Jennifer Myers, MD; Mayumi Nakagawa, MD, PhD
Phase 1 Clinical Trial of Intralesional Injection of Candida Antigen for the Treatment of Warts
Arch Dermatol. 2010;146(12):1431-1433. doi:10.1001/archdermatol.2010.350